All-in-one foam heel dressing
Mepilex Border Heel
Five-layer foam dressing for pressure ulcer prevention and management of heel wounds
Mepilex® Border Heel dressing is the only five-layer foam heel dressing with proprietary Deep Defense® Technology – proven to prevent pressure injuries when used in conjunction with other standard prevention protocols
The dressing can also be used for managing moderate- to high- exuding wounds of the heel area. It absorbs and retains exudate
The enhanced design still includes Safetac® – the original less-pain contact layer with silicone adhesion. So the dressing is comfortable and confirms to the patients anatomy
- Proven to prevent pressure injuriess – when used with other standard prevention protocols
- Proprietary Deep Defense Technology protects against the extrinsic forces - pressure, shear and friction; and manages micro-climate, the key factors responsible for pressure ulcers
- Mepilex Border with Safetac technology minimises pain during dressing changes
. - Good fluid handling capacity and retention
- Reduces risk of maceration
Mepilex® Border Heel dressing is the only five-layer foam heel dressing with proprietary Deep Defense Technology® – proven to prevent pressure ulcers when used in conjunction with other standard prevention protocols
The dressing can also be used for managing moderate- to high- exuding wounds of the heel area. It absorbs and retains exudate
This recently-launched enhanced design includes new handling tabs and thicker borders, allowing easier application and patient checking
The enhanced design still includes Safetac® – the original less-pain contact layer with silicone adhesion. So the dressing is comfortable and confirms to the patients anatomy
- Proven to prevent pressure ulcers – when used with other standard prevention protocols
- Proprietary Deep Defense Technology protects against the extrinsic forces - pressure, shear and friction; and manages micro-climate, the key factors responsible for pressure ulcers
- Mepilex Border with Safetac technology minimises pain during dressing changes
. - Good fluid handling capacity and retention
- Reduces risk of maceration
Further product information
When to use Mepilex Border Heel
Treatment
Mepilex® Border Heel is specifically designed to fit the heel. The five-layer absorbent foam structure effectively manages exudate and the wound environment. So you can use it in the treatment of exuding wounds such as pressure ulcers, diabetic foot ulcers, heel ulcers, traumatic wounds, and other secondary healing wounds.
Pressure ulcer prevention
Mepilex Border Heel has been proven to prevent pressure ulcers
Note: using dressings as a prophylactic therapy does not replace the need for a comprehensive pressure ulcer prevention protocol.
How to use Mepilex Border Heel
Watch how to easily apply Mepilex Border Heel to help you achieve the best result for your patient.
How Mepilex Border helps in pressure ulcer prevention
Discover how Mepilex Border Heel helps pressure ulcer prevention. It is easier than you may think.
Watch Prof. Gefen explain the modern science on aetiology of pressure ulcers
Downloads
Product details
| Ref. No. | Description | Pieces Per Inner | NPC Code | PIP Code |
| 282710 | Mepilex Border Heel 22 x 23 cm | 10 | ELA1019 | - |
| 283250 | Mepilex Border Heel 18.5 x 24 cm | 5 | ELA651 | 383-1617 |
Related products
'References'







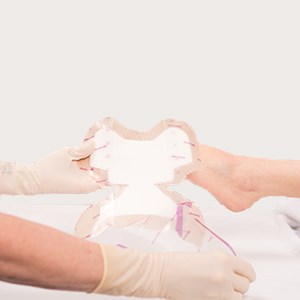 After the skin/wound is prepared, dry the area thoroughly. Then remove the central backing and slide the dressing under the heel to the desired location.
After the skin/wound is prepared, dry the area thoroughly. Then remove the central backing and slide the dressing under the heel to the desired location.
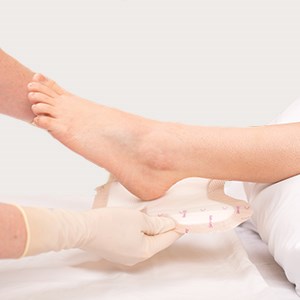 Apply the adherent part of the dressing marked ‘A’ (see picture above) to the posterior heel/ Achilles tendon areas, positioning the narrowest part of the dressing at the base of the heel. Do not stretch.
Apply the adherent part of the dressing marked ‘A’ (see picture above) to the posterior heel/ Achilles tendon areas, positioning the narrowest part of the dressing at the base of the heel. Do not stretch.
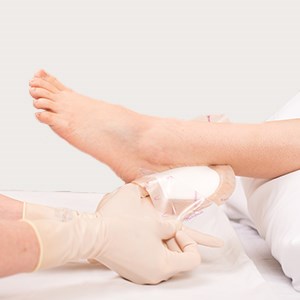 Remove the backing from one of the area ‘A’ flaps (ankle flaps). Apply and smooth. Repeat with the other side. Do not stretch.
Remove the backing from one of the area ‘A’ flaps (ankle flaps). Apply and smooth. Repeat with the other side. Do not stretch.
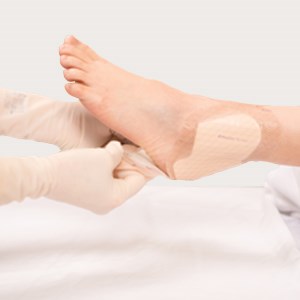 Gently apply the adherent part of the dressing marked ‘B’ (see illustration above) under the plantar surface of the foot. Do not stretch.
Gently apply the adherent part of the dressing marked ‘B’ (see illustration above) under the plantar surface of the foot. Do not stretch.
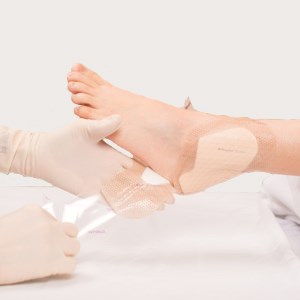 Remove the backing from one of the area ‘B’ flaps (flaps with tabs). Apply and smooth border. Repeat with the other side. Do not stretch.
Remove the backing from one of the area ‘B’ flaps (flaps with tabs). Apply and smooth border. Repeat with the other side. Do not stretch.
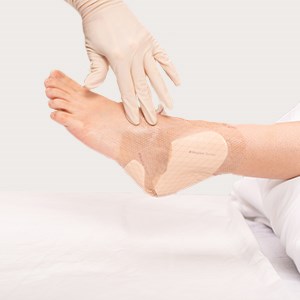 Press and smooth the dressing to ensure the entire dressing is in contact with the skin.
Press and smooth the dressing to ensure the entire dressing is in contact with the skin.
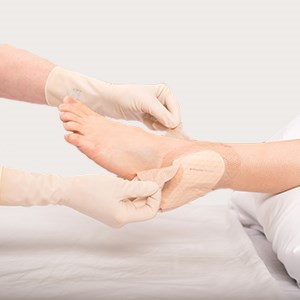 Gently pull handling tabs to release dressing from skin.
Gently pull handling tabs to release dressing from skin.
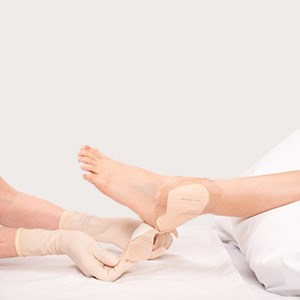 Continue to release the dressing from the skin using the handling tabs until the skin is exposed for skin check.
Continue to release the dressing from the skin using the handling tabs until the skin is exposed for skin check.
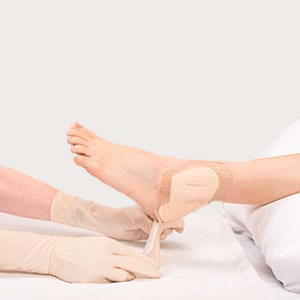 While maintaining the dressing position at the proximal edge of ‘A’ (see picture), perform assessment of the skin.
While maintaining the dressing position at the proximal edge of ‘A’ (see picture), perform assessment of the skin.
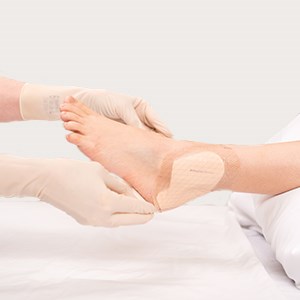 Re-apply the foam and border of the dressing. Make sure the flaps with the tabs are placed over the ankle flaps.
Re-apply the foam and border of the dressing. Make sure the flaps with the tabs are placed over the ankle flaps.
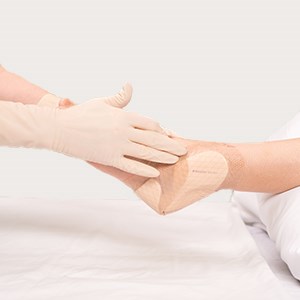 Confirm dressing is replaced to its original position, making sure the border is intact and flat.
Confirm dressing is replaced to its original position, making sure the border is intact and flat.
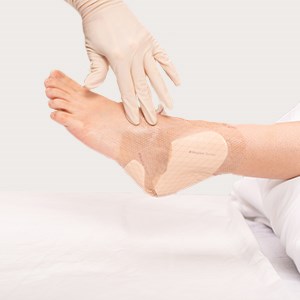 Press and smooth the dressing to ensure the entire dressing is in contact with the skin. Re-apply non-skid sock if using, especially for ambulatory patients.
Press and smooth the dressing to ensure the entire dressing is in contact with the skin. Re-apply non-skid sock if using, especially for ambulatory patients.








